MDA–Thai FDA Medical Device Regulatory Reliance Programme: Full Implementation from 1 May 2026 A landmark shift in ASEAN medical device regulation took effect on 1 May 2026, as the Medical Device Authority (MDA) of Malaysia and the Thai Food and Drug Administration (Thai FDA) of Thailand announced the full implementation of the MDA–Thai FDA Medical […]
Thai FDA Medical Device Applications: Why they Get Delayed or Rejected (and How to Fix It) Thailand offers a structured regulatory framework for medical devices, but in practice, a significant number of applications are delayed or rejected due to avoidable issues. Understanding the underlying causes is essential for companies planning a successful market entry. Many […]
Announcement of the FDA Regarding the Criteria, Procedures, and Conditions for Registration of Medical Device Manufacturing Facilities (2026) A seemingly “ordinary” title, but a regulatory shift On 15 January 2026, the Thai Food and Drug Administration (Thai FDA) issued a new Announcement on the Criteria, Procedures, and Conditions for Registration of Medical Device Manufacturing Facilities, […]
Factory License for Medical Devices in Thailand: Regulatory Requirements and Approval Pathway Establishing a medical device manufacturing facility in Thailand requires a specific regulatory authorization issued by the Thai FDA. The factory license is a prerequisite for any local production activity involving medical devices, regardless of classification. Unlike general business registrations or industrial permits, this […]
Thai FDA and the Expert Committee Pathway for Class 4 and Innovative Medical Devices The increasing role of the Thai FDA Expert Committee is reshaping the regulatory pathway for high-risk and innovative medical devices in Thailand. For manufacturers seeking approval of Class 4 products or technologies not yet recognized in reference countries, understanding how […]
Medical Device Grouping Criteria Under Thai FDA Regulations When applying for medical device registration in Thailand, one of the most important preliminary assessments concerns how products may be grouped under a single license. The Thai FDA does not automatically accept grouping requests submitted by applicants. The final determination regarding grouping structure rests exclusively with the […]
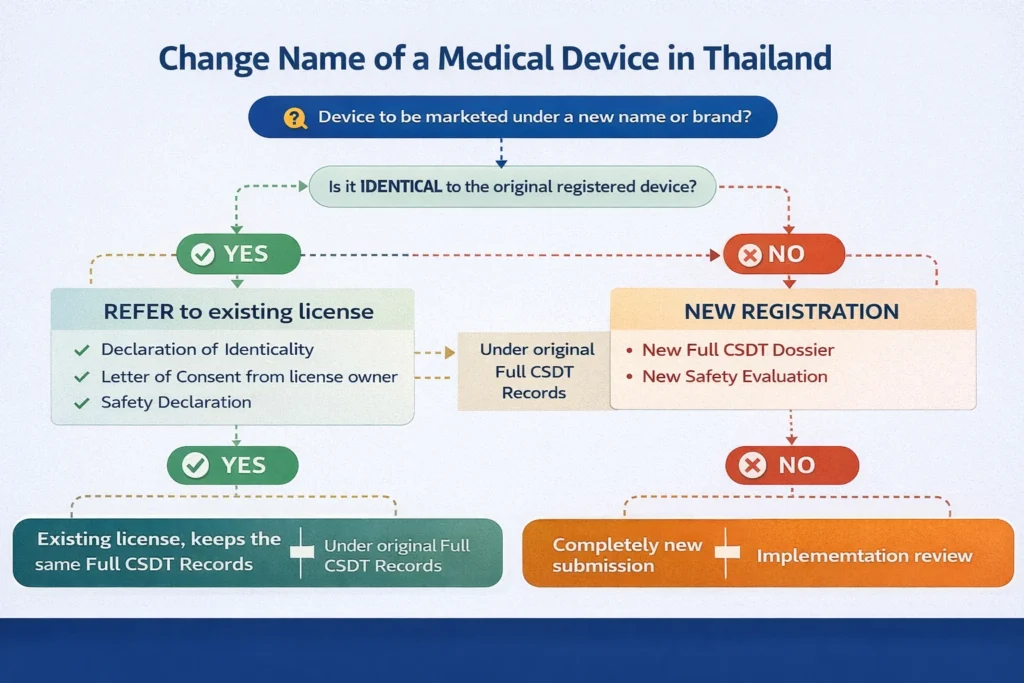
Change Name of a Medical Device in Thailand: Regulatory Pathway Using Refer The possibility to change name of a medical device in Thailand is often misunderstood by manufacturers and brand owners. Under Thai FDA practice, a name change is not automatically considered an administrative update. In many cases, changing the product name creates a new […]
Contact Lens Classification in Thailand: What Really Determines Risk Class Contact lenses are among the most misunderstood medical devices from a regulatory perspective. A large part of this confusion originates from the way contact lenses are marketed: daily, monthly, quarterly, semi‑annual, annual, disposable, extended wear, cosmetic, colored, etc. From a Thai FDA regulatory standpoint, these […]
Why Thai FDA Rejects Medical Device Registrations Every year, a significant number of Thai FDA medical device registration rejections and major delays occur, not because of defective products, but because of poorly prepared regulatory submissions. Medical device registration in Thailand is not a simple administrative filing. It is a structured regulatory project that requires correct […]
Foreign Business License for Medical Devices – Direct Import into Thailand An increasing number of foreign medical device manufacturers are reassessing their traditional market entry models for Thailand. Instead of appointing a local distributor or relying on a third-party License Holder, many manufacturers now aim to import and commercialize their products directly through a wholly […]
- 1
- 2