Medical Device Grouping Criteria Under Thai FDA Regulations When applying for medical device registration in Thailand, one of the most important preliminary assessments concerns how products may be grouped under a single license. The Thai FDA does not automatically accept grouping requests submitted by applicants. The final determination regarding grouping structure rests exclusively with the […]
Author Archives: Regulatory Team
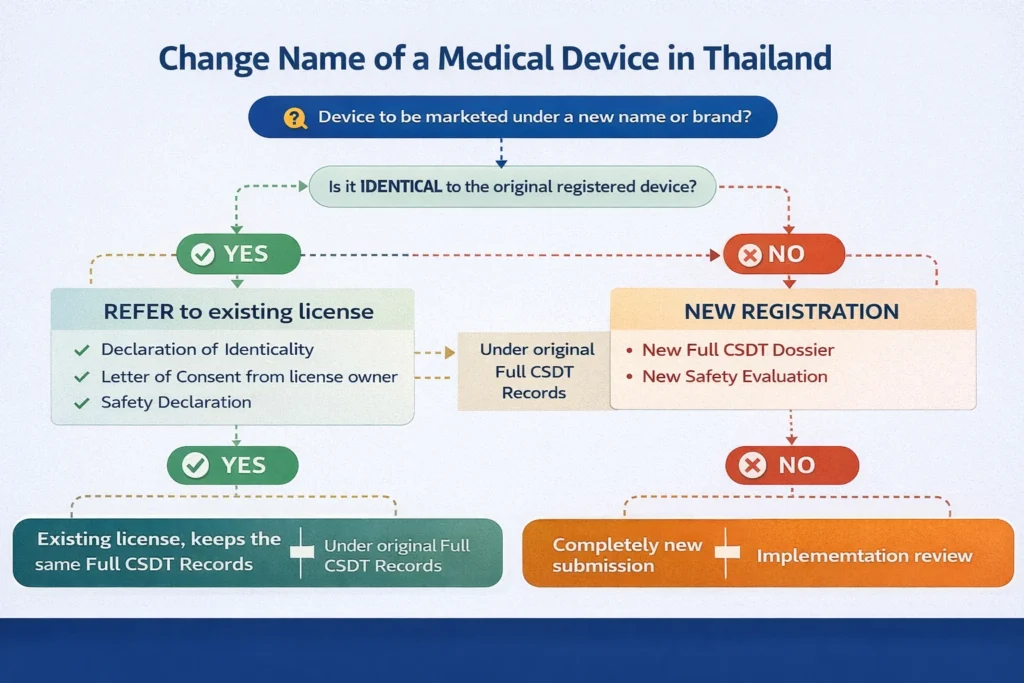
Change Name of a Medical Device in Thailand: Regulatory Pathway Using Refer The possibility to change name of a medical device in Thailand is often misunderstood by manufacturers and brand owners. Under Thai FDA practice, a name change is not automatically considered an administrative update. In many cases, changing the product name creates a new […]
Contact Lens Classification in Thailand: What Really Determines Risk Class Contact lenses are among the most misunderstood medical devices from a regulatory perspective. A large part of this confusion originates from the way contact lenses are marketed: daily, monthly, quarterly, semi‑annual, annual, disposable, extended wear, cosmetic, colored, etc. From a Thai FDA regulatory standpoint, these […]
Why Thai FDA Rejects Medical Device Registrations Every year, a significant number of Thai FDA medical device registration rejections and major delays occur, not because of defective products, but because of poorly prepared regulatory submissions. Medical device registration in Thailand is not a simple administrative filing. It is a structured regulatory project that requires correct […]
Foreign Business License for Medical Devices – Direct Import into Thailand An increasing number of foreign medical device manufacturers are reassessing their traditional market entry models for Thailand. Instead of appointing a local distributor or relying on a third-party License Holder, many manufacturers now aim to import and commercialize their products directly through a wholly […]
Medical Device Assembly in Thailand: Regulatory Challenges, Hidden Risks, and a Structural Loophole Thailand has become an increasingly attractive hub for medical device assembly, particularly for international manufacturers operating under BOI incentives, Free Trade Zones (FTZ), or Foreign Business License (FBL) structures dedicated to 100% export. This model is especially common among Chinese manufacturers seeking […]
Thai FDA GSDP Requirements for Medical Device Distribution in Thailand The Thai Food and Drug Administration (Thai FDA) has formally strengthened regulatory oversight over the distribution of medical devices by introducing Good Supply-chain Distribution Practices (GSDP) requirements. These obligations form part of a broader regulatory plan aimed at ensuring product integrity, traceability, and patient safety […]
Medical Device Importer of Record in Thailand In Thailand, the role of Importer of Record (IOR) for medical devices is often misunderstood or intentionally simplified.In reality, it is a non-delegable legal function that must be aligned with both regulatory authorization and fiscal responsibility. Any structure in which the Importer of Record: is different from the […]
Authorized Representative for Medical Devices in Thailand The Legal Role of the Authorized Representative Under Thai FDA Rules In the Thai medical device regulatory framework, the Authorized Representative (AR) is not a formal placeholder.It is the legal and fiscal cornerstone of market access. Any structure where the Authorized Representative: does not hold the licenses, does […]
Choosing a Medical Device License Holder in Thailand: Why Risk Matters More Than Price When entering the Thai medical device market, manufacturers often focus on obtaining a license as quickly and cheaply as possible.However, license holding in Thailand is not a one-time administrative step, but a long-term regulatory responsibility with direct legal and operational consequences. […]