Change Name of a Medical Device in Thailand: Regulatory Pathway Using Refer
The possibility to change name of a medical device in Thailand is often misunderstood by manufacturers and brand owners.
Under Thai FDA practice, a name change is not automatically considered an administrative update. In many cases, changing the product name creates a new marketed identity, which triggers additional regulatory obligations.
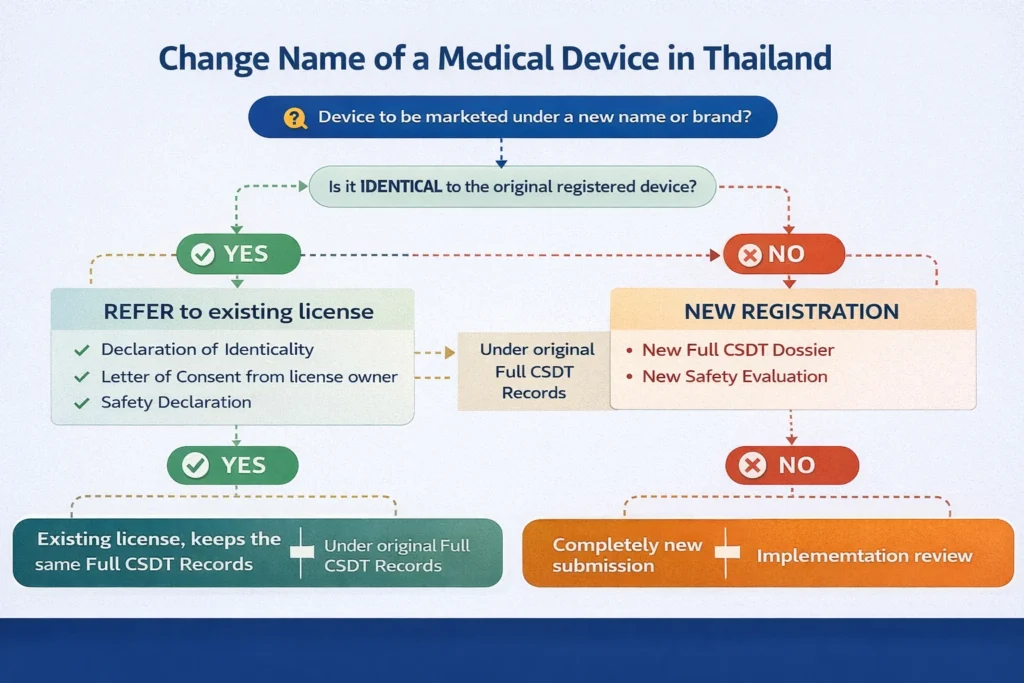
Thai FDA allows referencing an existing medical device registration dossier (Refer) when strict identity conditions are fulfilled.
Regulatory Basis for Change Name of a Medical Device in Thailand
Thai FDA allows applicants to reference an existing dossier when submitting a new application or notification under Full CSDT format.
When a company intends to change medical device’s name, Thai FDA evaluates whether:
-
The device remains technically identical
-
The change affects marketed identity
-
A new registration or notification is required
A change name of a medical device is therefore primarily a regulatory strategy question, not a simple labeling exercise.
When Refer Can Be Used to Change Medical Device Name
Refer may be applied to support a name change when:
-
The device has already been registered in Thailand
-
The new applicant has authorization from the original dossier owner
-
The device remains identical in all technical aspects
Refer does not eliminate Full CSDT requirements.
This operations without identity documentation is unlikely to be accepted.
Core Principle: Identity When You rebrand a Medical Device
Thai FDA focuses on identity, not similarity.
To support a change name, the applicant must demonstrate:
-
Identical design
-
Identical materials
-
Identical components
-
Identical manufacturing process
-
Identical intended use
-
Identical risk classification
If any of these differ, Refer is generally not appropriate.
Mandatory Documents for Change Name of a Medical Device (Refer Pathway)
When submitting a change name of a medical device using Refer, the following documents are required:
-
Declaration of Identicality for Refer
-
Letter of Consent for Refer
-
Safety Declaration
-
Full CSDT dossier
These documents must be uploaded individually in Thai FDA e-Submission.
New Name vs New Registration
Thai FDA practice shows that a change name of a medical device may be treated as:
-
A new registration supported by Refer
or -
A completely new standalone registration
There is no concept of “cosmetic” name change for medical devices.
Brand Strategy and Change Name of a Medical Device
If a manufacturer wishes to market the same device under multiple names:
-
Each name normally requires its own regulatory submission
-
Each submission must be supported by Full CSDT or Refer
Using multiple brand names without separate regulatory control is not acceptable.
Common Mistakes When Changing Name
-
Assuming change name of a medical device is minor
-
Attempting to update only labeling
-
Missing consent from dossier owner
-
Using Refer when technical differences exist
These mistakes frequently result in deficiency letters.
Conclusion: Changing the Name of a Medical Device Requires Regulatory Strategy
In Thailand, a change name of a medical device is a regulatory action, not a marketing decision.
Early assessment is essential to determine:
-
Whether Refer is acceptable
-
Whether a new registration is required
-
What documentation is needed