Medical Devices Regulatory Framework in Thailand
Scope of This Technical Resource
medicaldevice.co.th is an independent technical reference dedicated to the regulatory framework governing medical devices in Thailand.
The purpose of this website is to provide structured, neutral and technically accurate information on Thai FDA regulatory requirements, without offering commercial or consulting services.
The content published on this website is intended for manufacturers, regulatory professionals and industry stakeholders seeking a clear understanding of applicable rules, processes and compliance obligations.

Overview of Thai FDA Medical Device Regulation
Medical devices placed on the Thai market are regulated by the Thai Food and Drug Administration (Thai FDA) under a risk-based framework.
Regulatory requirements vary depending on device classification, intended use and technological characteristics.
Core regulatory areas include:
-
medical device classification
-
registration and approval procedures
-
appointment of a local license holder
-
post-market surveillance and vigilance
-
regulatory conditions for distribution
Correct interpretation of these elements is essential to ensure compliance and avoid regulatory delays.
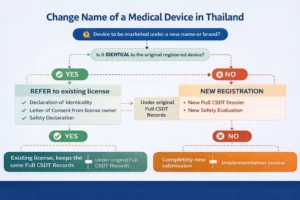
Medical Device Classification and Regulatory Impact
Thai FDA medical device classification determines the applicable regulatory pathway and directly affects documentation requirements, review timelines and post-market obligations.
Misclassification is a common source of regulatory inefficiency and may result in re-submissions or additional regulatory scrutiny during Thai FDA medical device registration.
Classification should therefore be assessed as an integrated component of the overall regulatory strategy rather than as a standalone administrative step.
Registration, License Holding and Local Accountability
Foreign manufacturers intending to commercialize medical devices in Thailand are required to appoint a local entity to act as license holder.
The Thai FDA license holder is legally responsible for:
-
regulatory submissions
-
compliance with Thai FDA requirements
-
post-market surveillance activities
-
regulatory communication with authorities
License holding represents a central element of the Thai regulatory system and carries defined legal and regulatory liabilities.
Post-Market Surveillance and Compliance Obligations
Post-market surveillance is a mandatory and ongoing requirement under Thai FDA regulations.
Obligations include adverse event reporting, field safety corrective actions and cooperation during inspections or audits.
These activities are typically coordinated by a local post-market compliance operator in Thailand, acting under the authority and responsibility of the license holder.
Effective post-market systems are critical to maintaining regulatory approvals and long-term market access.
Distribution Framework and Regulatory Alignment
Medical device distribution in Thailand is subject to regulatory conditions that are closely linked to license holding and post-market responsibilities.
Distribution models must ensure alignment between the distributor, the license holder and Thai FDA requirements.
In regulated environments, distribution activities are often implemented under a regulated medical device distribution model, designed to preserve compliance and accountability throughout the supply chain.
Regulatory Implementation and Market Entry Considerations
While this website focuses on technical and regulatory aspects, regulatory planning is frequently integrated into broader market entry strategies, particularly for manufacturers evaluating long-term presence, localization or investment in Thailand.
In such cases, regulatory requirements form a foundational component of a wider medical device market entry strategy in Thailand, where compliance, licensing and operational structure must be aligned.